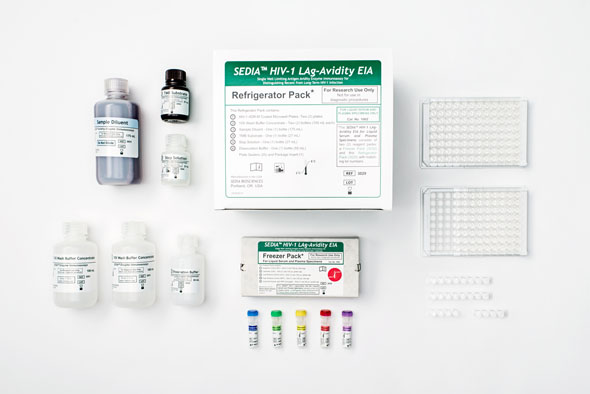
HIV LAB BASED PRODUCTS
AVAILABLE PRODUCTS
ADDITIONAL INFORMATION
Benefits of a Laboratory-Based Incidence Assay
Sedia® HIV-1 LAg-Avidity EIA allows for monitoring the outbreak of new infections in high-risk regions of the world.
High Throughput
Test multiple specimens at the same time.
Identify At-Risk Populations
Coordinate and monitor effectiveness of clinical studies.
Low False Recent Rate
Improved accuracy as published in the Recalibration of the Limiting Antigen EIA to Determine Mean Duration of Recent Infection in Divergent HIV-1 Subtypes study.
MDRI of 130 Days
Mean Duration of Recent Infection of approximately 130 days enables earlier detection of recent HIV-1 infections than other assays. Read More
CONTACT US
Want to know more or have any questions? Send us a message!